FDA announces changes to pain medication labels
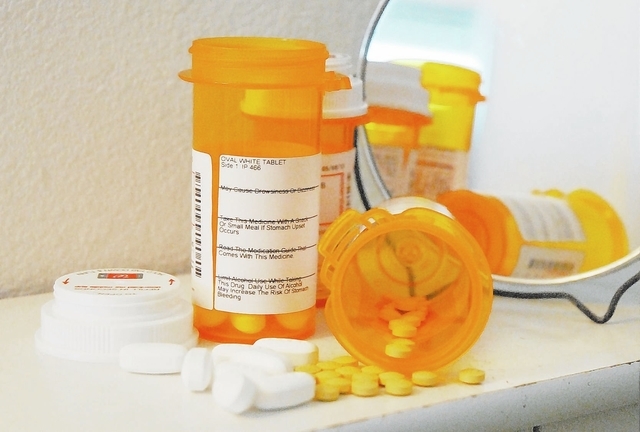
The U.S. Food and Drug Administration announced changes last week to safety label and post-market study requirements for extended-release and long-acting opioid pain medications in order to combat the rising problem of their misuse.
The changes are a result of the agency’s attempt to “combat the crisis of misuse, abuse, addiction, overdose, and death from these potent drugs that have harmed too many patients and devastated too many families and communities,” FDA Commissioner Dr. Margaret A. Hamburg said in a Sept. 10 statement.
“Today’s action demonstrates the FDA’s resolve to reduce the serious risks of long-acting and extended-release opioids while still seeking to preserve appropriate access for those patients who rely on these medications to manage their pain,” she said.
The new requirements for the prescription safety label will change current language included with the medications to state they should be used only for the management of pain “severe enough to require daily, around-the-clock, long-term opioid treatment and for which alternative treatment options are inadequate.”
According to the FDA the new labeling updates will further clarify that ER/LA opioid medications should be reserved for use in patients for whom other treatment options, such as non-opioid analgesics or immediate-release-opioids, are inadequate or do not provide sufficient pain management.
“The FDA’s primary tool for informing prescribers about the approved uses of medications is the product labeling,” Dr. Douglas Throckmorton, deputy director for regulatory programs in the FDA’s Center for Drug Evaluation and Research, said. “These labeling changes describe more clearly the risks and safety concerns associated with the ER/LA opioids and will encourage better, more appropriate, prescribing, monitoring and patient counseling practices involving these drugs.”
In addition to new labeling requirements, the FDA, after recognizing more information is needed to better assess the long-term affects and risks of taking ER/LA opioid medications, announced that it will also begin requiring drug manufacturers to conduct further post-market studies and clinical trials of these types of medications.
“The goals of these post-market requirements are to further assess the known serious risks of misuse, abuse, increased sensitivity to pain (hyperalgesia), addiction, overdose and death,” a press release from the FDA states.
Additionally the FDA announced it will be requiring manufacturers to to place a new warning label on medications cautioning users that chronic use of EL/LA opioids during pregnancy can result in neonatal opioid withdrawal syndrome or NOWS, which maybe be life-threatening and required management according to neonatal experts.
“The FDA remains committed to improving the safety of opioids and to continuing to engage in efforts to evaluate and mitigate the risks associated with opioid use, Dr. Throckmorton said in a statement. “Today’s safety labeling changes reflect the FDA’s current understanding of the risks and benefits of these products. The FDA will evaluate the results of the post-market studies, continue to monitor relevant safety data, and take further safety action, as warranted.”